- #CATHODE RAY EXPERIMENT JJ THOMPSON FULL#
- #CATHODE RAY EXPERIMENT JJ THOMPSON PORTABLE#
- #CATHODE RAY EXPERIMENT JJ THOMPSON SERIES#
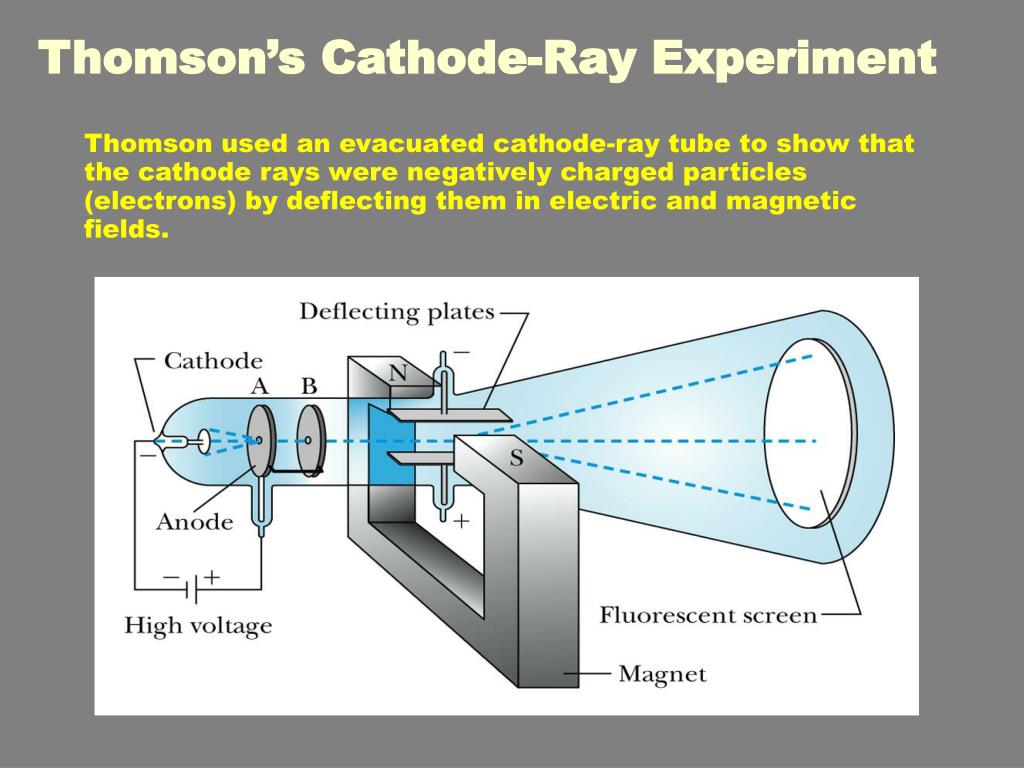
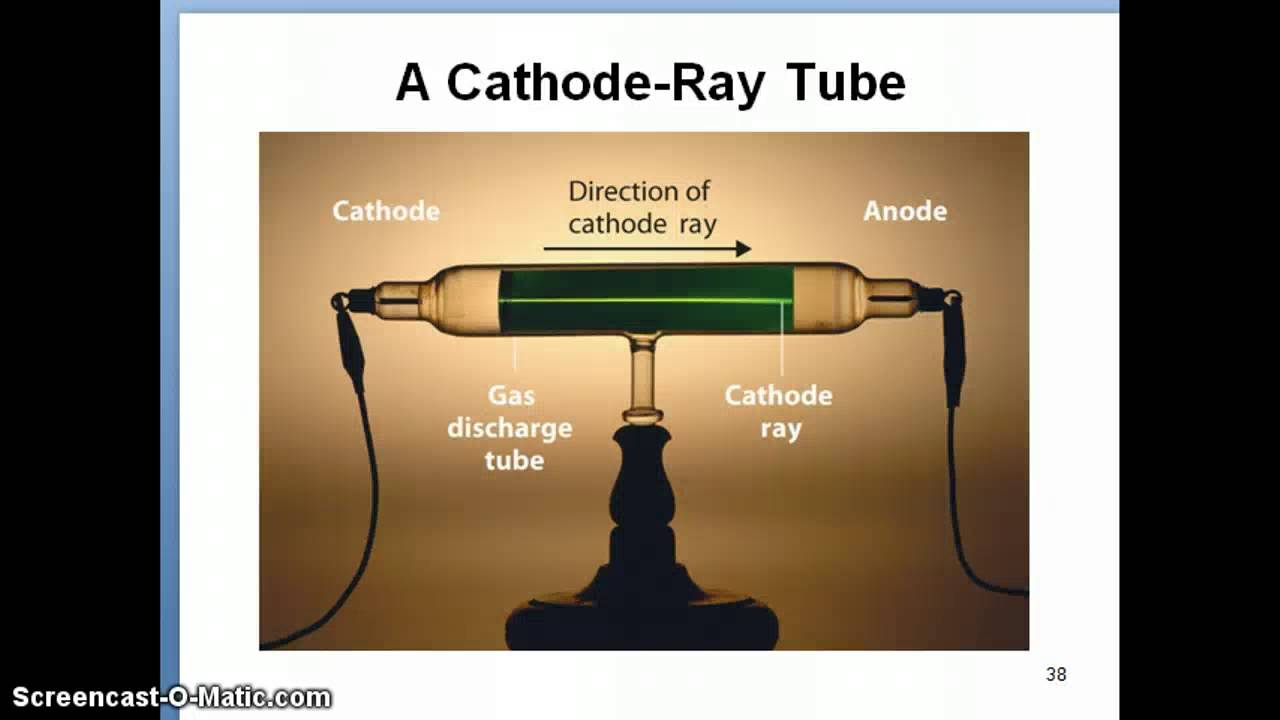
This experiment was performed using a cathode ray tube (Crooke’s tube). Thomson made a detailed study of the discharge of electricity through gases under very low pressure. Thompson, conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from. The above experiment should not be attempted except under the supervision of qualified laboratory personnel.See the related link below. Electrons were the first of sub-atomic particles to be discovered, by J.J. This is because the atmospheric pressure keeping the mercury up the tube is lower at a higher altitude.Note: Mercury is a dangerous substance and must be handled with care. Thomson, is one of the most well-known physical experiments, which led to the discovery.
#CATHODE RAY EXPERIMENT JJ THOMPSON PORTABLE#
The volume in the tube above the column of mercury will be filled with a vacuum.If you were to then take your portable barometer on a hike up a mountain, you would notice that the volume of the vacuum in the tube would become greater, and the column of mercury in the tube become shorter. Abstract: The Cathode Ray Tube (CRT) experiment performed by J. Some of the mercury will flow out of the tube into the bowl, but the air pressure will cause the remaining mercury remain in the tube up to a height of (about) 760 mm above the mercury level in the bowl. Then stand the inverted tube (with its sealed end in the bowl of mercury), and while the end of the tube is immersed in the mercury, remove the fitted seal, allowing the mercury trapped inside the tube to flow into the bowl.
#CATHODE RAY EXPERIMENT JJ THOMPSON FULL#
First, completely fill the tube with mercury, and temporarily close of the open end of the glass tube with a fitted seal, so the glass tube will be completely full of mercury and closed at both ends.
#CATHODE RAY EXPERIMENT JJ THOMPSON SERIES#
For much of his career, Thomson worked on various. J.) Thomson (18561940) performed a series of experiments in 1897 designed to study the nature of electric discharge in a high-vacuum cathode-ray tube, an area being investigated by many scientists at the time. Thomsons work on gaseous discharge, University of Bath, 1985, in which further details may be found. Thomson was the Cavendish professor of Experimental Physics at Cambridge University and director of its Cavendish Laboratory from 1884 until 1919. Thomson had shown that cathode rays behave as one would expect negatively charged material particles to behave. The original experiment that demonstrated this was a portable mercury column.If you have a large bowl full of Mercury (a liquid metal) and a 1 meter long glass tube open at one end, then you can make a simple barometer. Thomson, 1856-1940 see photo at American Institute of Physics) is widely recognized as the discoverer of the electron. How can you prove that the atmosphere exerts pressure? Thomson had discovered particles that were smaller than anything Dalton had imagined.
